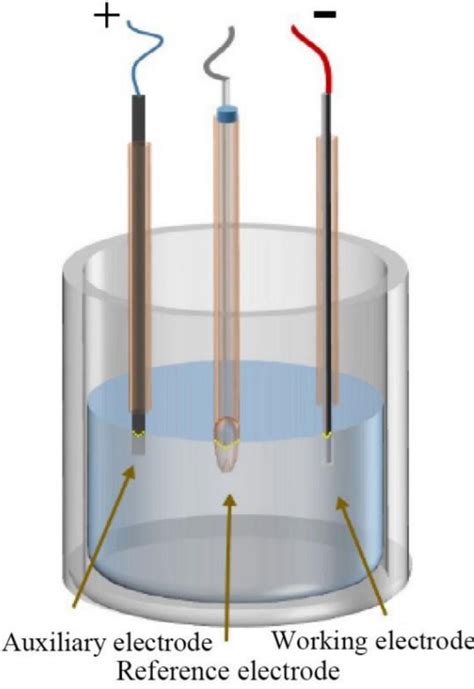
Also to know is, why do we use 3 electrodes?
In order to apply potential, we require some standard/reference electrode, whose potential is almost constant. So we have to avoid using this RE as current carrying electrode. So we require a third electrode called Counter or Auxiallry electrode and its main purpose is to complete the circuit to carry current.
Subsequently, question is, why three electrode system is better than two electrode system? Three-electrode setups have a distinct experimental advantage over two-electrode setups: they measure only one half of the cell. That is, the potential changes of the working electrode are measured independent of changes that may occur at the counter electrode.
In respect to this, what are the electrode types for three electrode cells?
In a traditional three-electrode cell, three different electrodes (working, counter, and reference) are placed in the same electrolyte solution.
How does a electrode work?
The electrode can either be an anode or a cathode. An anode receives current or electrons from the electrolyte mixture, thus becoming oxidized. When the atoms or molecules get close enough to the surface of the electrode, the solution in which the electrode is placed into, donates electrons.